 
In related terms, another unit of mass often used is Dalton (Da) or unified atomic mass unit (u) when describing atomic masses and molecular masses. Molecular mass or molar mass are used in stoichiometry calculations in chemistry. This Calculator has been tested on Internet Explorer version 6 only,įirefox might not show all fields correctly. For question or remarks please contact us. !!! Lenntech BV cannot be held responsible for errors in the calculation, Make sure you enter the molecule of crystallization at last (e.g. The calculator handles at most two different bracket levels. The molecular mass calculator will recognize the entered formula's, which are included in the list of organic compounds. Or you can choose by one of the next two option-lists, which contains a series of common organic compounds (including their chemical formula) and all the elements. This online calculator you can use for computing the average molecular weight (MW) of molecules by entering the chemical formulas (for example C3H4OH(COOH)3 ). Molecular Weight Calculator Molecular Weight Calculator Separation and Concentration Purification Request.Plant Inspection & Process Optimalisation.Thus, the standard atomic weight of argon, A r°(Ar) =, indicates that atomic-weight values of argon in normal materials are expected to be from 39.792 to 39.963.įor iridium, the standard atomic weight A r°(Ir) = 192.217 ± 0.002 indicates that atomic-weight values of iridium in normal materials are expected to be from 192.215 to 192.219.įor more information on the interpretation of the uncertainty please consult the recent IUPAC Technical Report by Possolo et al. The reported uncertainties of the standard atomic weights are such that the atomic-weight values of normal materials are expected to lie in the given interval with great certitude. (3) our ability to precisely determine the atomic masses of the isotopes.Įlements in the first category are distinguished by an interval standard atomic weight. (2) our ability to determine the isotopic abundances, and 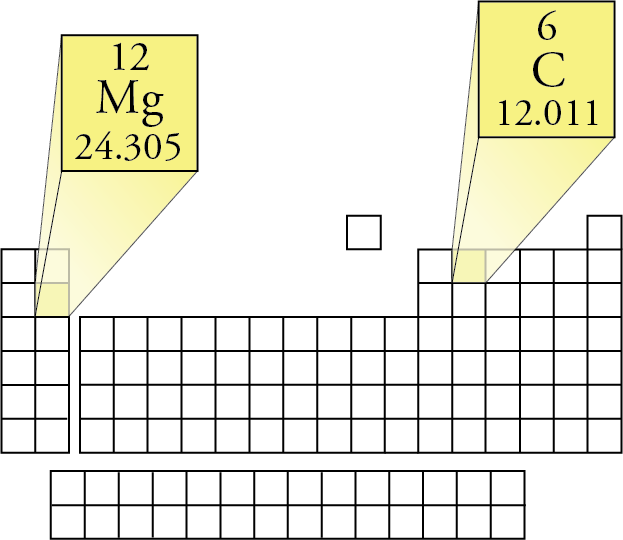
(1) well-documented natural variations of isotopic abundances, There are three broad groups of elements depending on what is the main cause of the uncertainty of their standard atomic weights: The most recent Standard Atomic Weights are presented in this Table and they are based on the "Atomic Weights 2021" report. R Range in isotopic composition of normal terrestrial material prevents a more precise standard atomic weight being given the tabulated value and uncertainty should be applicable to normal material. Substantial deviations in atomic weight of the element from that given in the table can occur. M Modified isotopic compositions may be found in commercially available material because the material has been subjected to some undisclosed or inadvertent isotopic fractionation. The difference between the atomic weight of the element in such materials and that given in the table may exceed the stated uncertainty. G Geological and biological materials are known in which the element has an isotopic composition outside the limits for normal material. The most recent definitive table of the standard atomic weights. Since 1902, the Commission regularly publishes critical evaluation of atomic weights of elements and below is Standard atomic weights are CIAAW recommended values for atomic weights applicable to all normal materials. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
